Description:
On July 17, 2025, the Infectious Diseases Society of America (IDSA) released its first-ever guidelines specifically addressing the management and treatment of complicated urinary tract infections (cUTIs). These guidelines provide practical recommendations for clinicians caring for both inpatient and outpatient populations with cUTI. The document expands upon prior UTI guidelines by offering a clinically relevant framework to distinguish uncomplicated from complicated infections, outlining a stepwise approach to empiric antimicrobial selection, and providing evidence-based recommendations regarding timing of intravenous-to-oral transition and optimal treatment duration.
Main Text
Urinary tract infections (UTIs) are among the most common bacterial infections, with heterogeneous clinical manifestations. While many uncomplicated UTIs (uUTIs) may resolve spontaneously or respond well to short-course antibiotic therapy, complicated UTIs (cUTIs) are associated with higher morbidity and an increased risk of therapeutic failure (1).
On July 17, 2025, the Infectious Diseases Society of America (IDSA) published its inaugural clinical practice guideline for the management and treatment of complicated urinary tract infections. This guideline provides updated definitions to differentiate uncomplicated (uUTI) from complicated (cUTI) infections, a structured stepwise approach to empiric antimicrobial therapy for cUTI, and consensus recommendations regarding the timing of intravenous-to-oral transition and the duration of therapy.
Key Highlights from the 2025 IDSA Guideline:
- Updated Classification of Uncomplicated (uUTI) and Complicated (cUTI) Infections
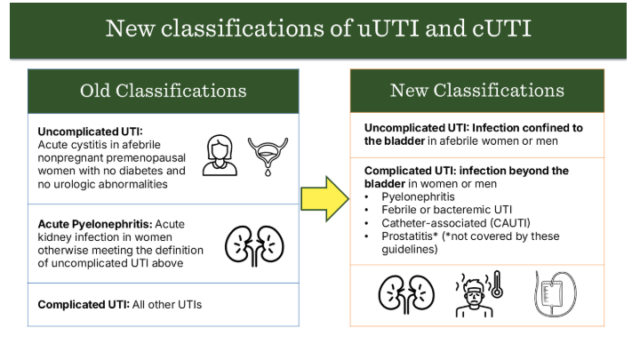
- The revised classification emphasizes the presence or absence of localizing or systemic signs (e.g., fever) suggestive of infection extending beyond the bladder.
- Readily identifiable clinical features available at the point of care (such as vital signs or the presence of an indwelling urinary catheter) are prioritized over factors that typically require urologic evaluation (e.g., anatomical abnormalities, urinary retention).
Figure 1.0. Comparison of prior versus updated classification criteria for uUTI and cUTI.

- Antimicrobial Therapy Selection for cUTI
2.1. Empiric Antimicrobial Therapy for cUTI
The guideline provides recommendations for empiric antimicrobial selection when complicated UTI (cUTI) is suspected. Agents are categorized into preferred versus alternative options according to disease severity. A structured four-step framework is emphasized to individualize therapy:
- Assess the severity of illness.
- Evaluate risk factors for antimicrobial resistance.
- Consider patient-specific characteristics.
- In cases with concurrent bacteremia, refer to susceptibility testing.
Once culture results are available, therapy should be de-escalated or transitioned to targeted antimicrobial treatment accordingly.
Selected IDSA 2025 Recommendations for Empiric Therapy in cUTI:
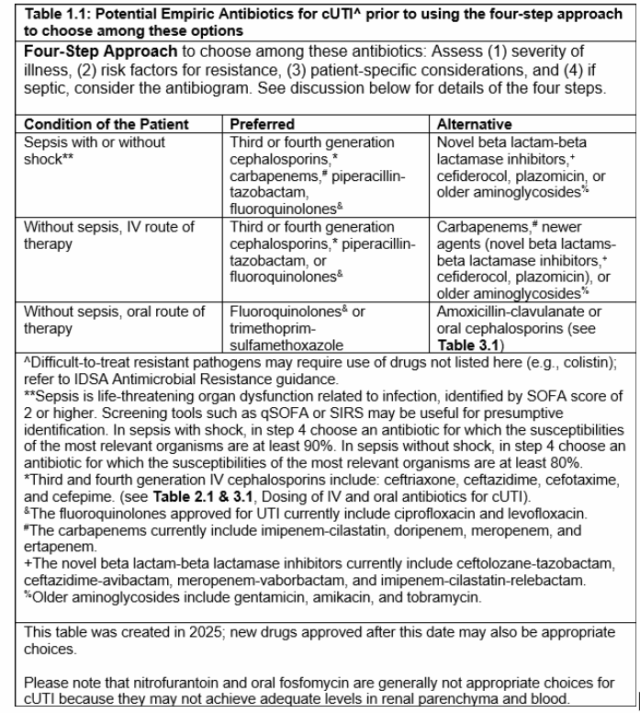
- Recommendation 1. For patients with sepsis secondary to cUTI, initial empiric therapy should be guided by the four-step framework (Figure 1.1). Preferred options include:
- Third- or fourth-generation cephalosporins,
- Carbapenems,
- Piperacillin–tazobactam, or
- Fluoroquinolones,
rather than newer β-lactam/β-lactamase inhibitor combinations, cefiderocol, plazomicin, or older aminoglycosides (conditional recommendation; very low to moderate certainty of evidence).
- Recommendation 2. For patients with suspected cUTI without sepsis, initial empiric therapy should also follow the four-step framework (Figure 1.1). Preferred agents include:
- Third- or fourth-generation cephalosporins,
- Piperacillin–tazobactam, or
- Fluoroquinolones,
rather than carbapenems, newer β-lactam/β-lactamase inhibitor combinations, cefiderocol, plazomicin, or older aminoglycosides (conditional recommendation; very low to moderate certainty of evidence).
A more detailed list of empiric antimicrobial options is provided in Table 1.1 of the guideline.
2.2. Stepwise Approach to Empiric Antimicrobial Selection for cUTI
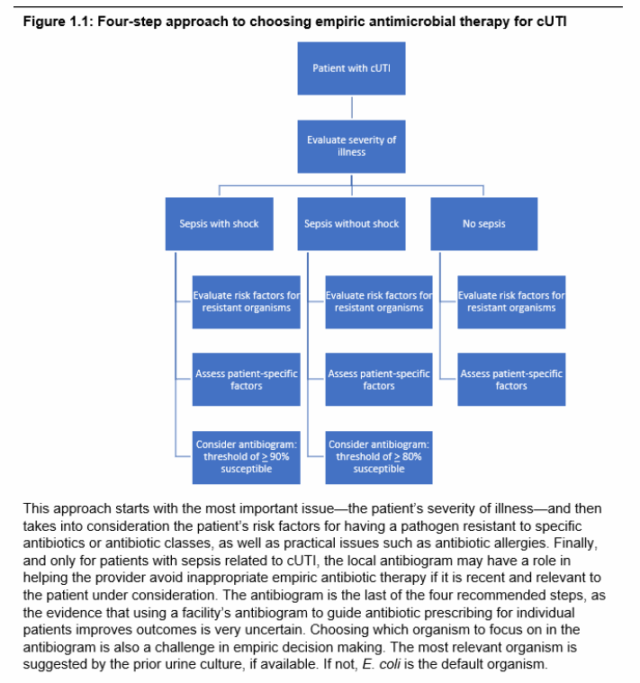
The guideline outlines a four-step process to optimize empiric antimicrobial selection in patients with suspected complicated urinary tract infection (cUTI):
- Assess the severity of illness to guide prioritization of initial empiric therapy.
- Evaluate individual risk factors for antimicrobial resistance.
- Incorporate patient-specific considerations (e.g., comorbidities, drug allergies, renal function) to minimize adverse events.
- If bacteremia is present, reference local antimicrobial susceptibility data to enhance treatment effectiveness.
Figure 1.1. Four-step framework for empiric antimicrobial therapy selection in cUTI.
2.3. Targeted Antimicrobial Therapy for cUTI
- Recommendation 3. In patients with confirmed cUTI, antimicrobial therapy should be narrowed to a targeted agent with definitive activity based on urine culture and susceptibility results as soon as available, rather than continuing empiric broad-spectrum therapy for the entire treatment course (conditional recommendation, low certainty of evidence).
2.4. Intravenous-to-Oral Transition in cUTI
- Recommendation 4. In patients with cUTI (including acute pyelonephritis) who demonstrate clinical improvement after initial parenteral therapy, an effective oral agent should be substituted in place of completing the entire treatment course intravenously (conditional recommendation, low certainty of evidence).
- Recommendation 5. In patients with complicated UTI (including acute pyelonephritis) accompanied by Gram-negative bacteremia who have improved clinically following initial parenteral therapy, oral step-down therapy may be considered if effective oral options are available, rather than completing the entire course intravenously (conditional recommendation, very low certainty of evidence).
2.5. Duration of Antimicrobial Therapy in cUTI
- Recommendation 6. In patients with cUTI (including acute pyelonephritis) who have achieved clinical improvement on effective therapy, shorter treatment courses are preferred: fluoroquinolones for 5–7 days (conditional recommendation, moderate certainty of evidence) or non-fluoroquinolone agents for 7 days (conditional recommendation, very low certainty of evidence), rather than prolonged therapy of 10–14 days.
- Recommendation 7. In patients with cUTI and concurrent Gram-negative bacteremia who show clinical response to effective therapy, shorter treatment durations (7 days) are suggested in preference to longer regimens (14 days) (conditional recommendation, low certainty of evidence).
For details, see the full IDSA 2025 Clinical Practice Guideline on Management and Treatment of Complicated Urinary Tract Infections (Executive Summary):
IDSA Guideline – Executive Summary (PDF)
Summary and Clinical Implications
The 2025 IDSA guideline provides clinically meaningful updates for physicians in selecting and optimizing antimicrobial therapy for cUTI. By emphasizing individualized empiric choices, timely de-escalation to targeted therapy, appropriate IV-to-oral transition, and shorter treatment durations, these recommendations aim to ensure effective and judicious antibiotic use in the context of rising antimicrobial resistance worldwide.
Although aminoglycosides are considered alternative options for empiric cUTI therapy, they remain clinically relevant—particularly in patients who fail to respond to, or cannot tolerate, preferred agents. Among them, plazomicin, a newer aminoglycoside, is favored over older agents such as gentamicin, tobramycin, and amikacin. However, given that plazomicin is not widely available in many regions, conventional aminoglycosides continue to be essential therapeutic options in real-world practice.
To support optimal management, Vinphaco provides high-quality injectable amikacin products, ensuring effective, safe, and cost-efficient treatment of complicated urinary tract infections:
- VINPHACINE (Amikacin sulfate 500 mg/2 ml)
- KA.NA.KACIN (Amikacin 500 mg/2 ml)
- AMIKATSIN (Amikacin 500 mg/2 ml)
- AMIKASIN-RV (Amikacin sulfate 500 mg/2 ml)
References:
- Broughton, E., Bektas, M., Colosia, A. et al. A Systematic Literature Review of the Epidemiology of Complicated Urinary Tract Infection. Infect Dis Ther 14, 1157–1181 (2025). https://doi.org/10.1007/s40121-025-01149-8
- https://www.idsociety.org/globalassets/idsa/practice-guidelines/cuti/executive-summary.pdf
- https://www.idsociety.org/practice-guideline/complicated-urinary-tract-infections/

 Myanmar
Myanmar