Description
Vinsalpium (salbutamol sulfate 2.5 mg/ipratropium bromide 0.5 mg/2.5 ml) is a short-acting bronchodilator in nebulized form, combining a β2-adrenergic agonist with an anticholinergic agent. Manufactured by Vinphaco (Vinh Phuc Pharmaceutical JSC) under international standards and approved for circulation in Vietnam, Vinsalpium was evaluated in the VINCOPD study for its short-term efficacy in COPD patients (both exacerbation and stable states) regarding clinical outcomes, lung function, and adverse events compared with the reference drug of the same active ingredients (Combivent – Boehringer Ingelheim). Results demonstrated that Vinsalpium was non-inferior to Combivent in efficacy and safety, with some advantages in symptom improvement and blood pressure stability. The VINCOPD study has been published in the Vietnam Medical Journal 2024, the Vietnam Pulmonology Association Journal, and presented at multiple national respiratory conferences.
Article Content
Chronic Obstructive Pulmonary Disease (COPD) is among the leading causes of disability and mortality worldwide. In Vietnam, ensuring effective, safe, and affordable treatment remains a top priority for clinicians. Vinsalpium (salbutamol sulfate 2.5 mg/ipratropium bromide 0.5 mg/2.5 ml) is a short-acting bronchodilator in nebulized form, combining a β2-agonist and an anticholinergic agent. It was developed, manufactured, and registered by Vinphaco (Vinh Phuc Pharmaceutical JSC) to provide a high-quality COPD treatment option while optimizing cost-effectiveness for Vietnamese patients.
To establish the quality, efficacy, and safety of Vinsalpium in COPD management, the VINCOPD study was conducted in 2023 by the Vietnam Pulmonology Association. The objective was to evaluate the short-term efficacy of Vinsalpium in COPD patients (during exacerbations and stable phases) in terms of clinical outcomes, lung function, and adverse effects compared with the reference drug of the same formulation (Combivent – Boehringer Ingelheim).
The VINCOPD study was carried out at five major hospitals and respiratory clinics nationwide, enrolling 260 COPD patients. Participants were randomized into two groups: Vinsalpium nebulization (n=130) and Combivent nebulization (n=130). Clinical symptoms and pulmonary function were assessed before and after treatment, and outcomes were compared between the two groups.
Patient characteristics: Among enrolled patients, 74.2% were managed with spirometry, while 3.8% were unmanaged. Frequent exacerbations were reported in 119 patients (45.8%), and 104 patients (40.0%) had at least one prior hospitalization for severe exacerbation. A total of 163 patients (67.2%) adhered to scheduled follow-ups. Prior treatment with at least one medication (including short-acting bronchodilators, long-acting bronchodilators, and corticosteroids) was documented in 213 patients (82.0%). The mean baseline Visual Analog Scale (VAS) score was 4.50. Thirty-three patients (12.7%) had evidence of old pulmonary tuberculosis lesions on chest X-ray. No significant differences in baseline characteristics were found between the two groups, except for a higher prevalence of old TB lesions in the Vinsalpium group (p=0.005).
Clinical and functional outcomes: Comparative analysis between the two groups showed that the efficacy and safety of Vinsalpium were non-inferior to Combivent. Minor advantages favoring Vinsalpium were observed in blood pressure stability before and after treatment (p=0.03), post-treatment VAS score (p=0.028), and VAS score obtained during telephone follow-up (p=0.013).
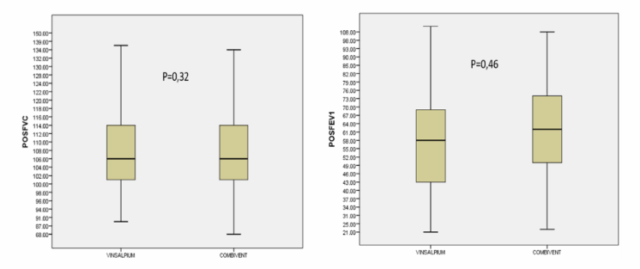
Figure 1. The Boxplot chart distributes the mean and SD values of %FVC pred. (left image) and %FEV1 pred. (right image) after using study and control drug
The VINCOPD clinical trial confirmed that the efficacy and safety of the investigational product (Vinsalpium) are non-inferior to those of the reference drug (Combivent). The study has been published in the Vietnam Medical Journal 2024, the Vietnam Pulmonology Association Journal, and presented at multiple national respiratory congresses.
This demonstrates Vinphaco’s commitment not only to delivering products that meet the highest quality standards but also to continuous innovation and the application of evidence-based medicine to substantiate the quality and effectiveness of its products in a clear and transparent manner.

Readers may access the full text of the VINCOPD study here:
https://tapchiyhocvietnam.vn/index.php/vmj/article/view/11909/10345

 Myanmar
Myanmar